 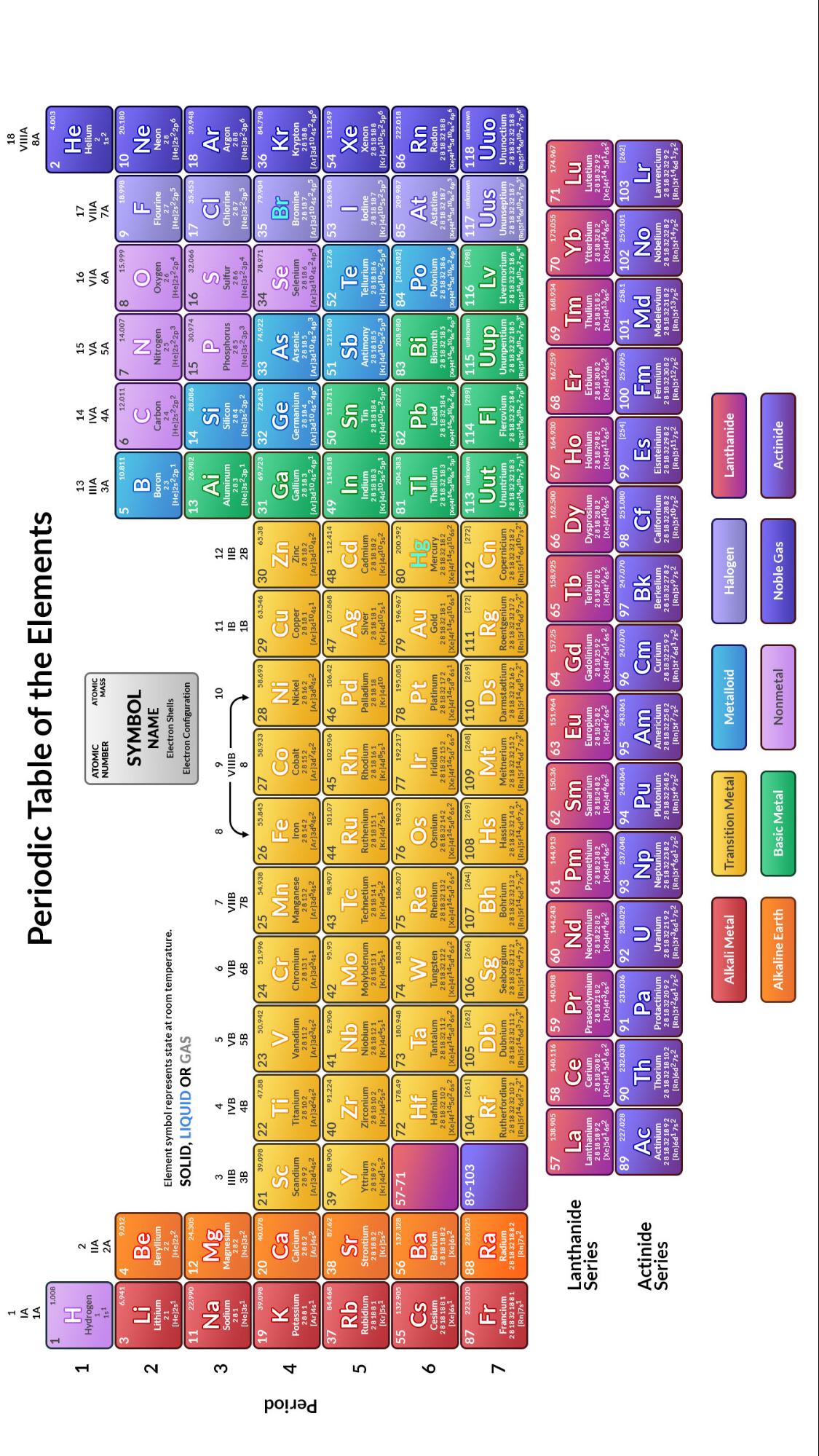 
The version shown below, in the International Union of Pure and Applied Chemistry (IUPAC) format, accounts for elements up to atomic number 118 and color-codes 10 different chemical series. The modern periodic table organizes elements according to their atomic numbers (number of protons in the nucleus) into 7 periods (vertical) and 18 groups (horizontal). The general consensus is that Mendeleev, not Meyer, was the true inventor of the periodic table because of the accuracy and detail of Mendeleev’s work.Įlement mendelevium (101) was named in honor of Dimitri Mendeleev.Įvolution of the Modern Periodic Table of Elements /GettyImages-758302997-5c215e3846e0fb0001b8bf7b.jpg)
German chemist Lothar Meyer was competing with Mendeleev to publish the first periodic table. You can read more about Mendeleev’s periodic table at the following link: The gaps represent undiscovered elements predicted by Mendeleev’s periodic table, for example, Gallium (atomic mass 69.7) and Germanium (atomic mass 72.6). Mendeleev’s periodic table consisted of Groups I to VIII in the modern periodic table. This first version of the periodic table is compared to the modern periodic table in the following diagram prepared by SIPSAWIYA.COM. Mendeleev observed that certain properties recur at regular intervals in the periodic table, thereby defining the groupings of elements. In 1869, Russian chemist Dimitri Mendeleev proposed the first modern periodic table of elements, in which he arranged the 60 known elements in order of their increasing atomic masses (average mass, considering relative abundance of isotopes in naturally-occurring elements), with elements organized into groups based their similar properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
